2,2-Diphenylglycine(Phenytoin RCA)
2,2-Diphenylglycine(Phenytoin RCA) Specification
- Molecular Formula
- C15H12N2O2
- Appearance
- White Solid
- Purity
- 98%
- Molecular Weight
- 252.268 Grams (g)
- Usage
- Phenytoin is used to prevent and control seizures (also called an anticonvulsant or antiepileptic drug). It works by reducing the spread of seizure activity in the brain.
2,2-Diphenylglycine(Phenytoin RCA) Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Payment Terms
- Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 500 Kilograms Per Month
- Delivery Time
- 3-4 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Main Export Market(s)
- Western Europe, Australia, Central America, Middle East, South America, Asia, Eastern Europe, North America, Africa
- Main Domestic Market
- All India
About 2,2-Diphenylglycine(Phenytoin RCA)
2,2-Diphenylglycine(Phenytoin RCA) pharmaceutical secondary standard, traceable to USP Synonym: 2,2-Diphenylglycine CAS Number 3060-50-2 Linear Formula H2NC(C6H5)2CO2H Molecular Weight 227.26 grade certified reference material packaging pkg of 100mg mp 245-247C (dec.)(lit.) pharmacopeia traceability traceable to USP 1535019 Analysis Note These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available. General description Pharmaceutical secondary standards for application in quality control, provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards.FAQs of 2,2-Diphenylglycine(Phenytoin RCA):
Q: What is the molecular formula of 2,2-Diphenylglycine (Phenytoin RCA)?
A: The molecular formula of 2,2-Diphenylglycine (Phenytoin RCA) is C15H12N2O2.Q: What is the molecular weight of 2,2-Diphenylglycine (Phenytoin RCA)?
A: The molecular weight of 2,2-Diphenylglycine (Phenytoin RCA) is 252.268 grams (g).Q: What is the purity level of 2,2-Diphenylglycine (Phenytoin RCA)?
A: The purity level of 2,2-Diphenylglycine (Phenytoin RCA) is 98%.Q: What does the appearance of 2,2-Diphenylglycine (Phenytoin RCA) look like?
A: The appearance of 2,2-Diphenylglycine (Phenytoin RCA) is a white solid.Q: What is the primary usage of 2,2-Diphenylglycine (Phenytoin RCA)?
A: Phenytoin is primarily used to prevent and control seizures. It works as an anticonvulsant or antiepileptic drug by reducing the spread of seizure activity in the brain.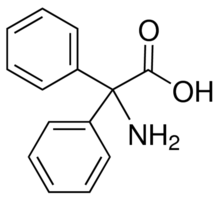

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Analytical Grade Chemicals Category
Octocrylene
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Weight : 361.48 Grams (g)
Purity : 98,0%
Appearance : yellow clear viscous liquid
Molecular Formula : C24H27NO2
Trifluridine
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Weight : 296.2 Grams (g)
Purity : 98%
Molecular Formula : C10H11F3N2O5
Trietazine
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Weight : 229.7098 Grams (g)
Purity : 98%
Appearance : Form: Liquid
Molecular Formula : C9H16ClN5
Haloperidol decanoate
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Weight : 530.1 Grams (g)
Purity : 98%
Appearance : White Solid
Molecular Formula : C31H41ClFNO3

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS