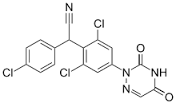
Azelastine Impurity D
Azelastine Impurity D Specification
- Usage
- Azelastine is a prescription medication used to treat allergy symptoms of the eyes or nose such as runny or stuffy nose, sneezing, itching.
- Purity
- 93%
- Molecular Weight
- 381.898 Carat
- Melting Point
- 225C
- Molecular Formula
- C22H24ClN3O
Azelastine Impurity D Trade Information
- Minimum Order Quantity
- 1 , , Kilograms
- Payment Terms
- Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 500 Kilograms Per Month
- Delivery Time
- 3-4 Week
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Main Export Market(s)
- Western Europe, Australia, Central America, Middle East, South America, Asia, Eastern Europe, North America, Africa
- Main Domestic Market
- All India
About Azelastine Impurity D
FAQs of Azelastine Impurity D:
Q: : What is the molecular formula of Azelastine Impurity D?
A: : The molecular formula of Azelastine Impurity D is C22H24ClN3O.Q: : What is the melting point of this impurity?
A: : Azelastine Impurity D has a melting point of 225C.Q: : What is the purity level of the product?
A: : The product has a purity level of 93%.Q: : What usage is associated with Azelastine and its related impurities?
A: : Azelastine is used to treat allergy symptoms, such as sneezing, itching, runny nose, and nasal congestion. Impurities like Azelastine Impurity D are instrumental in research and quality control processes for related formulations.Q: : Who benefits from using Azelastine Impurity D in their processes?
A: : Pharmaceutical suppliers, traders, and researchers working on Azelastine formulations benefit from incorporating Azelastine Impurity D into their processes.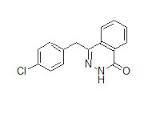

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Analytical Grade Chemicals Category
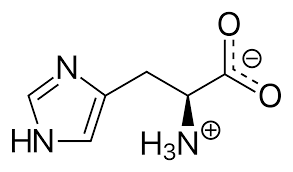
Histidine
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Formula : C6H9N3O2
Molecular Weight : 155.1546 Grams (g)
Melting Point : >243°C (dec.)
Bacopaside I
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Formula : C46H74O17
Molecular Weight : 899.1 Grams (g)
Appearance : Powder
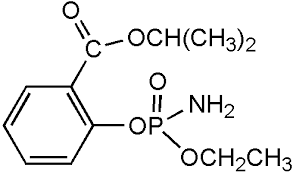
Isofenphos solution
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Formula : C15H24NO4PS
Molecular Weight : 345.4 Grams (g)
Melting Point : 2.9 C (37.2 F; 276.0 K)
Appearance : Clear colorless liquid
Diclazuril
Price 100 INR
Minimum Order Quantity : 1 Kilograms
Molecular Formula : C17H9Cl3N4O2
Molecular Weight : 407.64 Grams (g)
Melting Point : 290.5°C
Appearance : Off white

 Send Inquiry
Send Inquiry


 Send Inquiry
Send Inquiry Send SMS
Send SMS Call Me Free
Call Me Free