Pyrophosphoryl Chloride
Price 100 INR/ Kilograms
Pyrophosphoryl Chloride Specification
- Flash Point
- Not applicable (decomposes)
- CAS No
- 13498-00-3
- Physical State
- Liquid
- Ph Level
- Not applicable (acidic nature)
- Size
- Customizable, as per requirement
- Boiling point
- 131-135 C
- Shelf Life
- 24 months in original sealed packaging
- Melting Point
- -32 C
- Purity
- 98%
- Packaging Type
- Drum
- Molecular Weight
- 218.28 g/mol
- Usage
- Laboratory Reagent, Industrial Catalyst
- Molecular Formula
- Cl4O3P2
- Density
- 1.67 Gram per cubic centimeter(g/cm3)
- Storage Instructions
- Store in tightly closed container, in a cool, dry, and well-ventilated place, away from moisture
- Grade
- Industrial Grade
- Type
- Chemical Compound
- Application
- Pharmaceutical intermediates, Dye manufacturing, Chemical synthesis
- Appearance
- Colorless to pale yellow fuming liquid
- Purity(%)
- 98%
- Solubility
- Reacts violently with water
- Sensitivity
- Moisture sensitive
- Handling Precautions
- Use with adequate ventilation and wear suitable protective equipment
- Refractive Index
- 1.470
- HS Code
- 28129090
- Stability
- Stable under recommended conditions
- Odor
- Pungent irritating odor
- Incompatibility
- Water, alcohols, bases, strong oxidizing agents
- Transport Information
- Hazardous, UN No. 3265, Corrosive Liquid
Pyrophosphoryl Chloride Trade Information
- Payment Terms
- Cash Against Delivery (CAD), Cash in Advance (CID)
- Main Export Market(s)
- Middle East, Asia, Africa
- Main Domestic Market
- All India
About Pyrophosphoryl Chloride
Pyrophosphoryl Chloride
Diphosphoryl chloride, Pyrophosphoric tetrachloride,
Mol Formula Cl4O3P2
CAS No. 13498-14-1
EC No. 236-824-4
Molecular Weight 251.76
Key Applications
Pyrophosphoryl Chloride is integral to the manufacture of pharmaceutical intermediates, dyes, and other chemicals. Its high reactivity and purity make it valuable for processes that require precise chemical transformations, such as laboratory reagent use and industrial catalysis.
Safe Handling and Storage
The compound must be handled with adequate ventilation and protective equipment. It is stable under recommended conditions, but reacts violently with water, alcohols, and bases. Storage in a cool, dry, well-ventilated place, away from moisture, ensures its longevity and safety, with a shelf life of 24 months in sealed packaging.
Transport and Packaging
Classified as a hazardous, corrosive liquid (UN No. 3265), Pyrophosphoryl Chloride is shipped in drums with customizable sizes as per client requirements. Adherence to international transport regulations is essential to prevent incidents and maintain product integrity.
FAQ's of Pyrophosphoryl Chloride:
Q: How should Pyrophosphoryl Chloride be stored for optimal stability?
A: Pyrophosphoryl Chloride should be stored in a tightly closed container, in a cool, dry, and well-ventilated area, away from moisture and incompatible substances such as water, alcohols, and strong oxidizers to maintain stability and prolong shelf life.Q: What protective measures are needed during the handling process?
A: Handling requires adequate ventilation and the use of appropriate personal protective equipment, including gloves, goggles, and protective clothing, due to its corrosive properties and pungent, irritating odor.Q: When is Pyrophosphoryl Chloride typically utilized in industry and research?
A: The compound is frequently used during the synthesis of pharmaceutical intermediates, dye manufacturing, and chemical research as a reagent or industrial catalyst due to its high reactivity and purity.Q: Where does Pyrophosphoryl Chloride find major applications?
A: Its primary applications are in chemical laboratories, the pharmaceutical industry, and dye manufacturing facilities, where precise chemical synthesis and transformation are required.Q: What are the benefits of using Pyrophosphoryl Chloride in chemical synthesis?
A: Its high purity and potent reactivity facilitate efficient chemical transformations, making it ideal for processes demanding rigorous control and accuracy, such as those in pharmaceutical and dye synthesis.Q: How is Pyrophosphoryl Chloride transported safely to customers?
A: It is classified as a hazardous, corrosive liquid and shipped under UN No. 3265 regulations, typically in drums. Transport requires compliance with safety guidelines to prevent exposure and material degradation.Q: What should be done in case of accidental exposure to moisture?
A: Immediate isolation of the substance and removal from moisture sources is essential. Evacuate the area and ensure ventilation, as it reacts violently with water. Consult the safety data sheet (SDS) for emergency actions and seek professional help if needed.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Laboratory Chemicals Category
5 Bromo 5 nitro1 3dioxane
Price 100 INR / Kilograms
Minimum Order Quantity : 10 Kilograms
Purity(%) : >99%
Appearance : White to offwhite crystalline powder
CAS No : 69898459
Grade : Reagent Grade
HMB Calcium
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : 99%
Appearance : White crystal powder
CAS No : 135236725
L-Phenylalanine Amino Acid
Price 100 INR / Kilograms
Minimum Order Quantity : 1 Kilograms
Purity(%) : 90%
Appearance : White or white crystalline powder
CAS No : 63912
Grade : Food
liquid 1,2-Dichloroethane
Price 100 INR / Pack
Minimum Order Quantity : 1 Pack
Purity(%) : 98%
Appearance : Colorless liquid
CAS No : 107062
Grade : Special Grade

 Send Inquiry
Send Inquiry
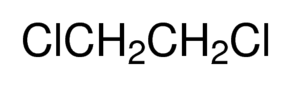


 Send Inquiry
Send Inquiry Send SMS
Send SMS